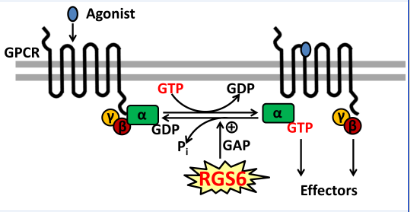
GPCR Signaling:

Regulation of G protein-coupled receptor (GPCR) signaling by Regulator of G protein signaling (RGS) proteins. RGS proteins function as GTPase-activating proteins for specific Ga subunits and thereby function to terminate G protein signaling by GPCRs.
mRNA splicing:
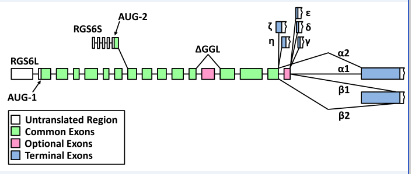
Diagram of the complex splicing of human RGS6 pre-mRNA to generate 36 splice variants. Two primary transcripts encode the 5′-splice forms of RGS6; the AUG-1 start site produces a transcript that encodes the RGS6L forms of the protein while the AUG-2 start site produces a transcript that encodes the RGS6S forms of the protein. Retention or removal of exon 13 (first pink square) generates transcripts that encode for proteins containing or lacking a complete GGL domain, respectively. 3′-splicing generates transcripts containing seven distinct 3′ exons. RGS6 α and β transcripts exist in two forms that arise from either the retention (α1 and β1) or removal (α2 and β2) of exon 18 (second pink square). Image adapted from Chatterjee et al, J. Biol. Chem. 2003.
Protein isoforms:
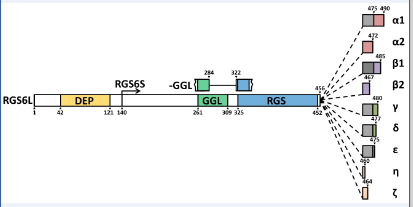
Predicted protein structure of human RGS6 proteins. There are predicted to be numerous RGS6 protein isoforms that differ in length due to the following: inclusion or exclusion of the disheveled EGL-10, pleckstrin homology (DEP) domain at their N-terminus, inclusion or exclusion of a complete G gamma subunit-like (GGL) domain, and the inclusion of one of seven distinct C-termini. RGS6 proteins with either the long or the short N-terminus are labeled as RGS6L or RGS6S, respectively. The C-terminal domains are labeled as α, β, γ, δ, ε, η, and ζ. The α and β C-termini exist in two forms, either with (α1 and β1) or without (α2 and β2); an 18 amino acid sequence encoded by exon 18 (grey square) of the RGS6 gene. Finally, proteins that lack the GGL domain are designated as −GGL proteins. Amino acid numbers are included to specify where key regions of the protein begin and end. Image adapted from Chatterjee et al, J. Biol. Chem. 2003.
RGS6 protein model:
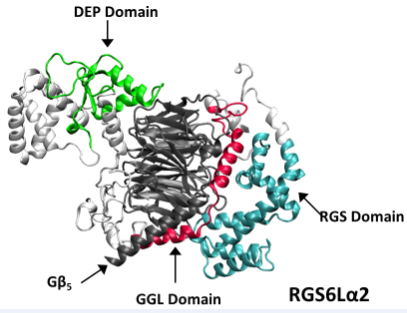
Structural model of RGS6La2 in complex with Gb5 based upon the known structure of the RGS9-Gb5 complex (PDB ID: 2PBI). Model was generated by Dr. Koland (Univ. of Iowa).
RGS patho:
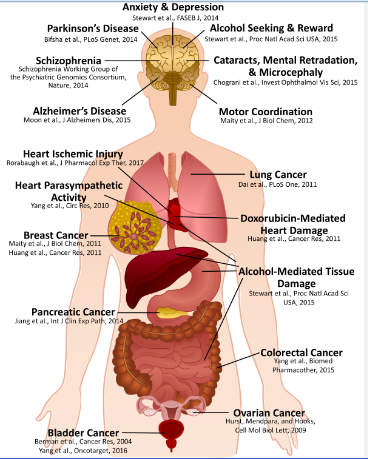
Schematic outlining identified physiological and pathophysiological functions of RGS6.